
You have to heat a mixture of hydrogen and iodine constantly to get it to react, but the reaction is reversible - so all you get is a mixture of hydrogen, iodine and hydrogen iodide formed. Reactions of bromine and iodine with hydrogenĪ bromine and hydrogen mixture will give a mild explosion if you put a lighted taper in it. The two gases react together to make white fumes of solid ammonium chloride. The test used for hydrogen chloride is to expose it to ammonia gas from concentrated ammonia solution. It reacts with water vapour in the air to form a fog of hydrochloric acid droplets. Hydrogen chloride is a colourless poisonous gas which looks steamy in moist air. In the lab, you have to be very careful if you mix gas jars of hydrogen and chlorine to keep them well away from sunlight.īut you can also get them to react more gently if you lower a jet of burning hydrogen gas into a gas jar of chlorine. Hydrogen chloride gas is produced explosively when the mixture is exposed to light. Reaction of chlorine with non-metals: hydrogenĬhlorine also reacts with several non-metals, but the only one you are likely to meet at this level is the reaction with hydrogen. If you pass chlorine over heated iron wool in a tube, it also burns - but this time you need to heat it to make the reaction start. In an earlier video you have seen that iron wool burns in fluorine without the need to heat it, and you have also seen a more convincing video of iron burning in a gas jar of chlorine. It certainly shows that the iodine doesn't react as vigorously as the bromine because it scarcely glows. Note: This last video doesn't actually show this change in vigour all that well because it isn't doing all the reactions under the same conditions. You are most likely to come across the reaction with chlorine, but might also be expected to know that the reactions become less vigorous as you go down the group. The bromine one is similar.įor reasons we will look at later on this page, iodine only converts the iron to iron(II) iodide, FeI 2.Īt this level, you are unlikely to need to know this. You have just seen the equation for the chlorine and iron reaction. The reactions tend to get less vigorous as you go from chlorine to bromine to iodine. The next video repeats the reaction of iron with chlorine, but extends it to include the reactions with bromine and iodine as well. The red-brown compound formed when iron reacts with chlorine is iron(III) chloride, FeCl 3. There are two iron chlorides, iron(II) chloride and iron(III) chloride. This is masked by the reaction between lithium and the glass producing black solids. The video skips over the fact that lithium chloride is a white solid. I include it just because it is visually interesting. 
It is unlikely you would ever be asked about this reaction. 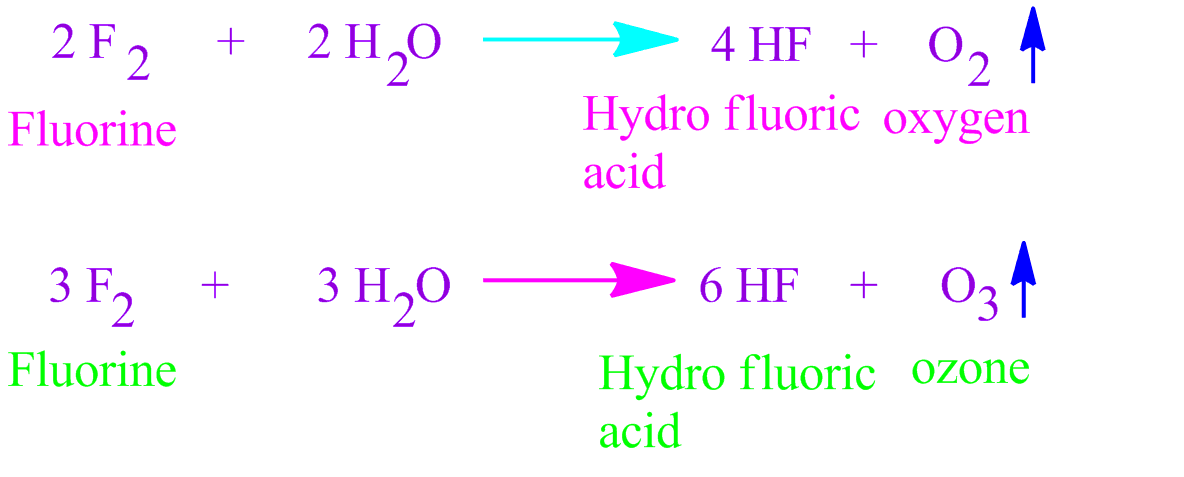
This is a reaction in which a lot of heat is given off. The video (and several others below) uses the phrase "highly exothermic reaction". The next most reactive element in the group is chlorine and we will start with that. You have just seen how vigorously fluorine reacts with almost all elements. I include it simply because it is interesting. You are unlikely to need this for exam purposes. There is an interesting bit of video from the University of Nottingham Periodic Table series which shows that I am not alone. 
Fluorine is extremely reactive and I have never even seen it, let alone used it. This is something you are very unlikely to see in the lab.īefore we go any further, I want to get the fluorine problem out of the way. It starts by producing yellow/green chlorine gas and then condensing this to a yellow/green liquid. This next short video shows chlorine, bromine and iodine. We can, however, predict what its properties might be by exploring the trends in the group. FĪstatine is very radioactive and short-lived. Group 7 is also known by its more modern name of Group 17. This page introduces the Halogens in Group 7 of the Periodic Table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
